No, this isn’t the kind of protein wrap you make yourself for lunch … or would even want to make for lunch! It’s a wrap-up of all that deep information I shared the last few weeks on the effects that amyloid and tau proteins have on cognitive decline in Alzheimer’s disease (AD) patients… and an introduction to the next generation of thinking in the battle against AD.
I thought I’d have fun with the title and cover image, I hope you like it. 😎
Let’s quickly recap what we know about AD disease progression and treatments presently …
The Present: Anti-amyloid monoclonal antibody treatments
Two “disease modifying” treatments are available to patients today, and they are lecanemab (marketed as Leqembi™) and donanemab (marketed as Kisunla™). Treatments referred to as disease modifying are ones that are proven to slow the disease’s progression not just mask the symptoms as they get worse.
Two more promising disease modifying treatments are in clinical trials, and they are remternetug and trontinemab. Remternetug just complete its phase 3 trial in March as had applied for FDA approval last year, and trontinemab is about halfway through its phase 3 trial.
All four treatments focus solely on either preventing amyloid plaque from forming or aggressive plaque removal. Each may have an indirect impact on tau formation by removing amyloid protofibrils or plaque which triggers tau toxicity, but that is not their focus.
The Near Future: There is a rapidly expanding pipeline of tau‑directed treatments and combination strategies. These fall into several categories:
Tau‑targeting therapies - The next frontier of treatments do one of three things: limit tau production altogether, attempt to prevent tau from becoming toxic, or remove tau after it becomes toxic.
Microtubule‑Stabilizing Therapies - Since amyloid and tau both destabilize microtubules, stabilizers like lithium, may protect the shared binding site and preserve neuronal structure.
Combination and Stage‑Specific Therapies - The future likely involves cocktail‑like treatment combinations for amyloid and tau, or stage‑specific therapy based on how far a person’s AD has progressed.
Research is increasingly showing that amyloid-beta and tau proteins are not independent villains but partners in a spiraling, destructive sequence that I call the amyloid–tau “dance of neuron death”.
Amyloid acts upstream, becoming toxic, clumping and displacing tau from microtubules in neurons.
Tau then becomes toxic, spreads between neurons, and causes cell death.
If that’s not enough, toxic tau can also amplify amyloid toxicity, creating a self‑reinforcing cycle.
Think of it this way, amyloid is the trigger, and tau is the bullet that actually kills neurons.
It all sounds so clear and definitive. At least that’s what we have heard for the last decade as the dominant theory of AD progression... If we stop amyloid from becoming toxic, we prevent it from triggering tau to do the damage, and we can stop AD from progressing.
Sadly, it’s not nearly that simple, there are many paths to cognitive decline in AD, and the pharmaceutical corporations may be pouring R&D into a far too narrow a treatment vector. Amyloid and tau proteins are neither the beginning nor the end of the AD progression, and that’s why we see that anti-amyloid treatments only modestly slow the disease’s progression. AD isn’t just about plaques and tangles, it’s about a brain under siege from many vectors. Each vector provides part of the story of how cognition fades and a person’s identity struggles to hold its ground.
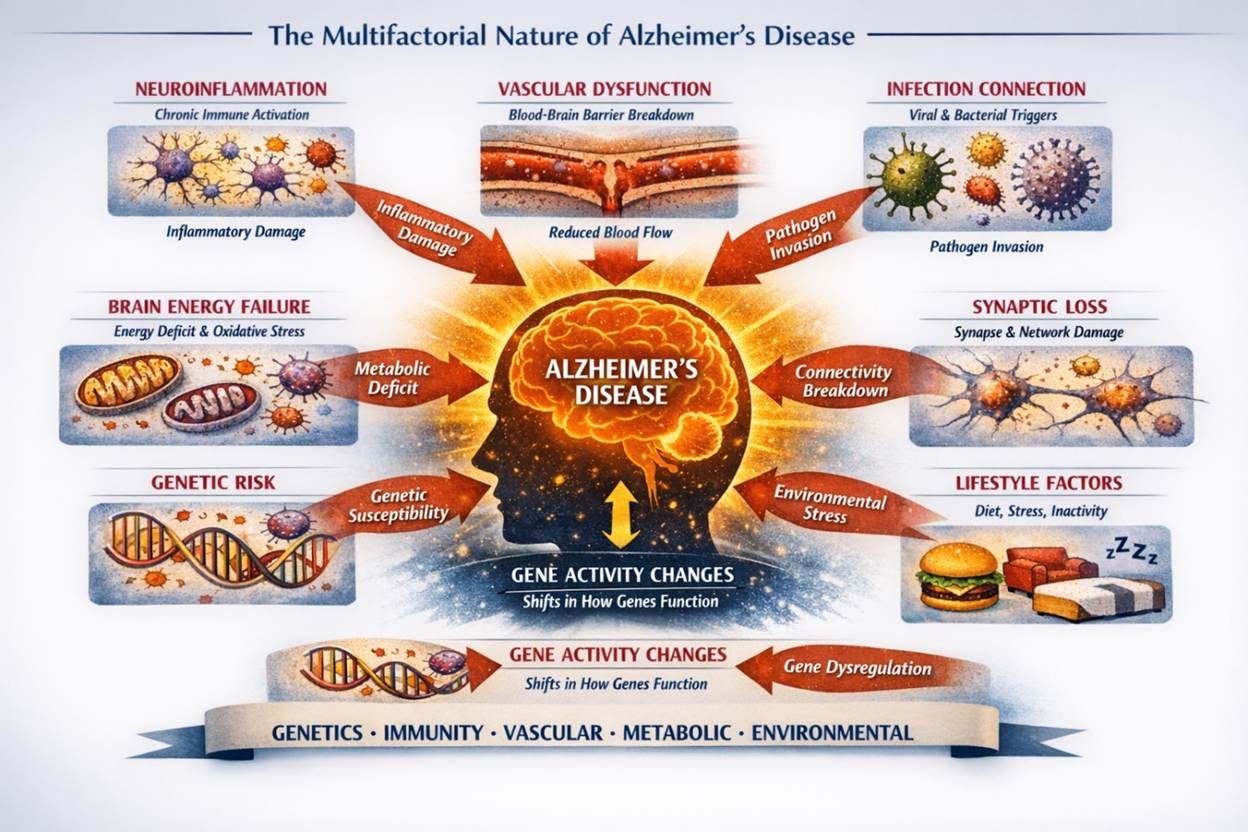
In the scientific community, Alzheimer’s is increasingly understood as a multi‑system failure, where amyloid and tau are important but sit within a broader ecosystem of the disease. Finally, new areas are being explored more seriously…
Neuroinflammation begins when the brain’s immune cells, meant to protect the brain, stay activated for too long. They flood the environment with inflammatory signals, damaging the very neurons they’re meant to defend.
Vascular Dysfunction quietly starves the brain. When blood vessels stiffen or leak, oxygen and nutrients can’t reach neurons, and waste products linger. As a result, the brain’s energy, repair, and cleanup processes falter.
Infection Connection reminds us that the brain doesn’t live in isolation. Chronic or latent infections, either viral or bacterial, may provoke immune responses that accelerate damage, turning defense into destruction. This is why scientists believe that vaccines for other illnesses like shingles, influenza, and pneumococcal are emerging as a significant way to prevent or slow the disease.
Brain Energy Failure captures the exhaustion at the heart of the disease. Our brains use over 20% of all our energy produced, so when neurons lose their ability to utilize energy efficiently, it leaves them vulnerable to oxidative stress and unable to sustain memory and thought.
Genetic Risk shapes vulnerability long before symptoms appear. Genes like APOE4 influence how the brain handles fats, repairs cells, and responds to inflammation, which sets the stage for resilience or risk.
Gene Activity Changes show that Alzheimer’s isn’t just about damaged genes, but about how they’re expressed. Over time, the brain’s “software” shifts, turning protective genes down and stress genes up.
Synaptic Loss is where the disease becomes personal. Connections between neurons, the basic component of memory and personality, begin to fray. Communication slows, then stops altogether.
Lifestyle Factors remind us that the environment we live in and how we treat our bodies matters! Sleep, exercise, diet, pollution, and stress all shape the brain’s resilience. These are the levers we can pull without waiting for treatments to develop, it’s the place where we should all feel a sense of urgency to address in our lives. I dove deep on this in prior articles covering the “four pillars of prevention” & “what might cause AD.”
Together, these forces converge into Cognitive Decline and Neurodegeneration, but they also point toward hope of a future where treatment means restoring balance across systems, not chasing a single vector or symptom.
This multi-pathway siege is why single‑target therapies (amyloid only, tau only) have limited impact, and why I believe multi‑modal strategies are the future.
Alzheimer’s is not a single‑pathway disease, and its treatment will not be a single‑pathway solution.
Coming Next: Our Broken Healthcare System
Peace my Friends and Thanks for Reading!
Note: What I’m sharing in my deep dive newsletter articles is based on personal research, but please remember I am not in the healthcare profession, and I have no credentials in that area past or present. I’m sharing what I’ve learned, and will continue to learn, in my own desperate effort to advocate for myself when meeting with my care team. I believe everything I share to be correct or I wouldn’t share it, but you should not take anything I say as medical advice.